|
Due to the existence of 2 lone pairs of oxygen atoms in ethers, they are able to dissolve in cold concentrated acids to form oxonium salts, which are stable. Therefore, at room temperature and pressure, they are gases, and they have a low boiling point. Ether: R1-O-R2Įthers are hydrophobic and unlike alcohols, they are only proton acceptors. The products of this are alkene and water. The loss of the whole hydroxyl group is called a dehydration reaction. If the hydrogen atom is not only donated but also replaced, an ether (R1-O-R2) is formed. In such a reaction, which is an oxidation reaction, an aldehyde is formed (if secondary alcohol reacts). įor this reason, the hydrogen atom can be easily donated. Thus, it is amphoteric as it can simultaneously be a weak base and a weak acid. This group can both donate and accept protons. As a result, reactions with electron-deficient molecules occur more easily. The extra electrons on the oxygen atom also make the hydroxyl group nucleophilic. This is also a polar group and enhances hydrogen bonding in organic compounds.Īlcohols are therefore normally in the liquid state and have high boiling points. As the oxygen atom has higher electronegativity, the electron pair from the water hydrogen atom is attracted to it, creating a negative charge. The hydroxyl group has hydrophilic properties. They can have names ending in -diol, -triol, etc. Compounds with 1, 2, or several hydroxyl groups are known as monovalent, divalent, or polyvalent alcohols. Primary and secondary alcohols are differentiated based on the number of organic molecules attached to the hydroxyl group. 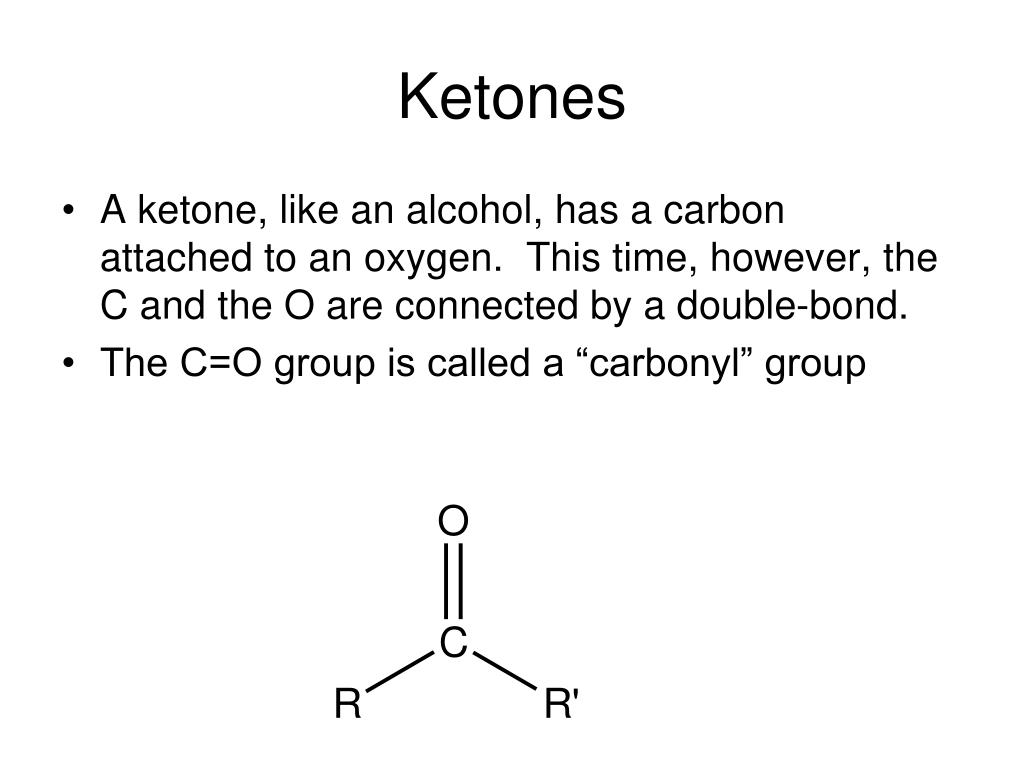
Phenols are benzene rings with an -OH group. In enols, the carbon atom with the hydroxyl group is connected to the neighboring carbon atom with a double bond. Organic compounds with a hydroxyl group are called alcohols. Image: ‘Hydroxy Group Structural Formula’ by Jü.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
